Multiple Approaches in Pharma Contract Manufacturing Explained

For OTC pharma and consumer health portfolios, contract manufacturing is not merely a capacity solution; it is a strategic operating model that integrates development, formulation, scale-up, and GMP manufacturing. For OTC pharma companies, using contract manufacturing enables faster market access, regulatory alignment across regions, and cost efficiency without the need for extensive in-house infrastructure. As […]
Benefits of Pharma Contract Manufacturing for Efficient Scale-Up

Pharma leaders face constant pressure to deliver products on time while controlling costs and meeting regulatory expectations. Internal manufacturing constraints, capacity gaps, and volatile demand can quickly turn into delays, budget overruns, and approval risk. Each decision carries commercial consequences, from missed launch windows to strained stakeholder confidence. The risk of locking into the wrong […]
Optimizing Chemical Reactions in Drug Manufacturing

Reaction optimisation services play a critical role in pharmaceutical development by improving process robustness, regulatory readiness, and manufacturing predictability. In a tightly regulated environment, optimised reactions are essential for achieving consistent yields, controlled impurity profiles, and reliable performance from lab scale to commercial production. For pharmaceutical developers and CDMO partners, reaction optimisation goes beyond laboratory […]
Comprehensive Guide to Stability Chamber Testing for Reliable Drug Development

Stability chambers testing has become a core pillar of pharmaceutical development, playing a direct role in defining shelf life, securing regulatory approval, and supporting long-term commercial success. In today’s globally regulated environment, generating reliable stability data is essential to demonstrate that a drug product remains safe, effective, and compliant throughout storage and distribution. For pharmaceutical […]
How Packaging Design Influences Drug Stability During Distribution
Ensuring drug stability through effective packaging is a critical challenge for pharmaceutical and biotech companies. Delays in development, regulatory setbacks, and commercial risks can all stem from inadequate packaging strategies. Misalignment between formulation, stability data, packaging, and regulatory expectations can drive up costs, extend timelines, and create avoidable compliance risk. Packaging must support the stability […]
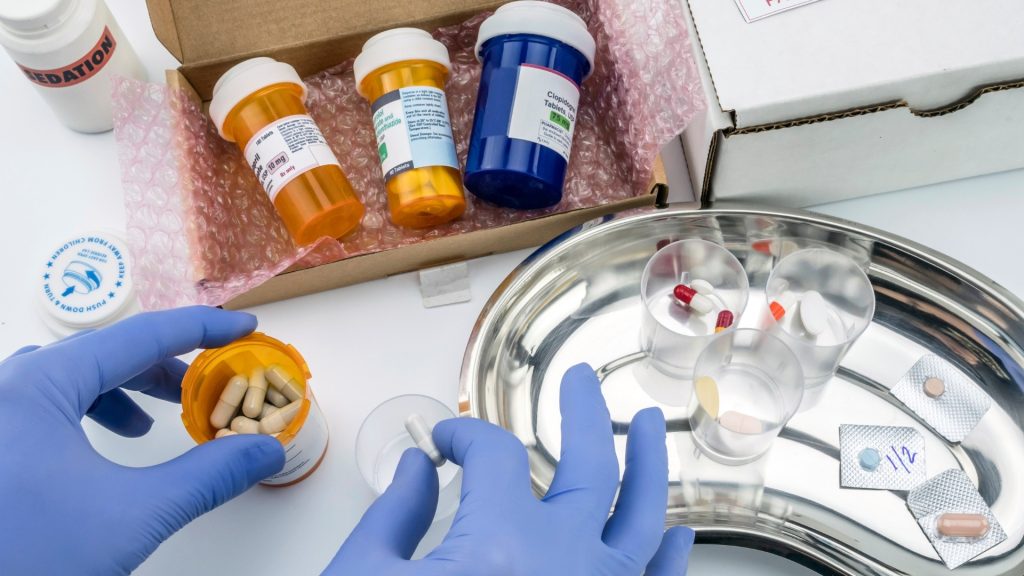
Comprehensive Drug Contamination Testing to Ensure Safety

Contamination events in pharmaceutical manufacturing routinely surface as late-stage regulatory findings, supply interruptions, or unplanned remediation costs with direct portfolio impact. As contamination can occur at any stage of the drug development and manufacturing process, from raw material sourcing to final packaging, the stakes are high. Contaminated drugs pose a direct threat to patient safety, […]
Achieving Operational Excellence in Pharmaceutical Production

Pharma operational excellence often breaks down after scientific promise is proven. Scale-up exposes weak assumptions. Technology transfer introduces variation. GMP readiness arrives late. Timelines stretch, costs climb, and regulatory confidence erodes. This leads to lost momentum, missed launch windows, deferred revenue, fragile supply plans, and growing pressure from boards, partners, and global health stakeholders to […]
Semi-Solid Dosage Forms: Production Methods and Benefits
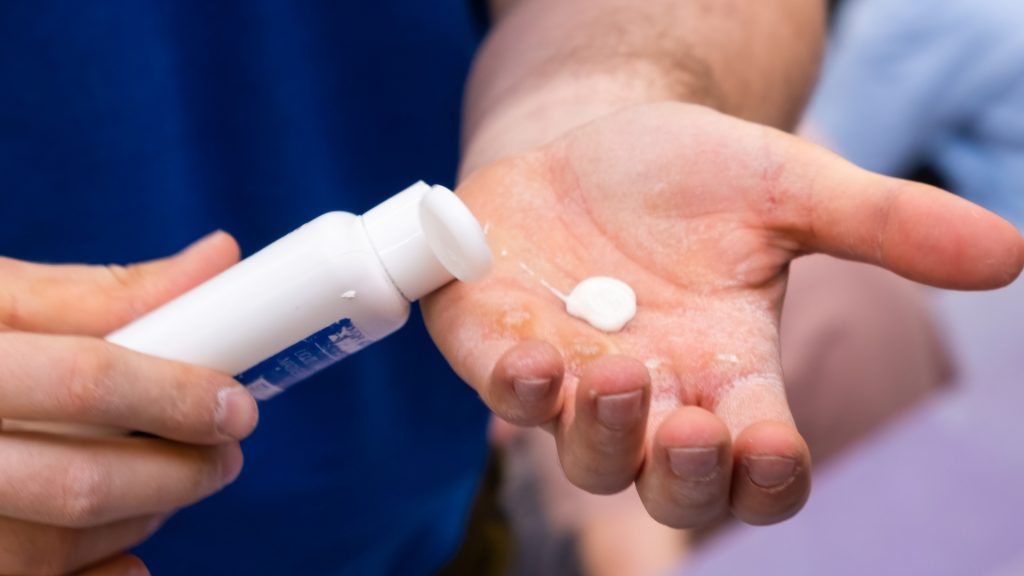
Semi-solid dosage forms often appear straightforward during early planning, yet they carry a high risk of late-stage disruption. Small changes in formulation or process conditions can quickly lead to stability concerns and scale-up failures that later attract regulatory scrutiny. These issues typically surface when timelines are compressed and commercial expectations are already locked. The challenge […]
Developing Inhalation Dosage Forms: Formulation & Device Fit

Inhalation dosage form development looks straightforward on paper. In reality, it is one of the fastest ways for a pharma program to lose time, money, and regulatory momentum. Small formulation missteps, poor alignment between formulation and selected device platforms, or late-stage comparability issues can push timelines out by years and place entire portfolios at risk. […]
Biosimilar Manufacturing: Unique Process and Analytical Needs

The biosimilars market is expanding rapidly, driven by the expiration of biologic patents and the increasing demand for affordable treatment options. However, manufacturing biosimilars presents significant challenges, including complex regulatory requirements, maintaining product consistency, and controlling production costs. As competition intensifies and regulatory scrutiny grows, pharmaceutical companies face mounting pressure to scale biosimilars production efficiently […]
