Packaging of drugs is not an isolated downstream activity; it is a core component of Chemistry, Manufacturing, and Controls (CMC) strategy that directly affects stability, regulatory acceptance, and supply continuity. As pharmaceutical portfolios shift toward complex formulations, global clinical programs, and multi-market commercialization, packaging decisions increasingly influence development risk and lifecycle cost.
From a strategic perspective, packaging supports regulatory compliance, supply chain resilience, and patient usability across diverse markets. It enables safe distribution under varying climatic and logistical conditions, particularly in multi-country programs where transportation, storage, and handling challenges are more pronounced.
This blog examines the role of drug packaging across primary, secondary, and tertiary levels, outlining key material, regulatory, and operational considerations and highlighting how integrated CDMO support helps ensure scalability, compliance, and market readiness.
Key Takeaways
- Packaging of drugs directly protects stability, safety, and usability, making it a critical quality and compliance driver.
- Early packaging strategy alignment reduces risk, preventing stability issues, relabeling, and supply chain disruptions.
- Clear primary, secondary, and tertiary packaging design ensures control, supporting compliance and efficient global distribution.
- Material compatibility and integrity testing are essential, as E&L, CCI, and barrier performance impact shelf life and patient safety.
- CDMO expertise enables scalable, regulatory-ready packaging, supporting consistent quality from clinical development through commercialization.
Why Packaging of Drugs Is More Critical Than Ever in Global Pharma?
Global pharmaceutical development now operates under compressed timelines, multi-country regulatory oversight, and increasingly fragile supply chains. In this environment, packaging of drugs functions as a risk-control layer rather than a downstream logistics activity.
Effective packaging strategies support:
- Product Stability Across Markets: Packaging systems must protect formulations from mechanical stress, moisture ingress, light exposure, and temperature variability, particularly when products move across diverse climatic zones and extended logistics routes common in emerging and regulated market supply chains.
- Regulatory and Data Integrity: Labelling, serialisation, and container-closure performance are integral to regulatory submissions and inspection readiness. Packaging failures often surface as compliance findings rather than operational issues.
- Distribution and Supply Continuity: Poorly designed packaging increases the likelihood of transit damage, relabeling, temperature excursions, and batch rejections, each of which can disrupt clinical programs or commercial supply.
Proper packaging helps:
1. Preserve Drug Quality
Packaging safeguards drugs against physical damage, microbial contamination, moisture, light, and temperature fluctuations. This is especially critical in multi-country clinical trials.
Example: Temperature-controlled containers keep biologics and vaccines stable during international shipping.
2. Communicate Accurate Information
Packaging conveys dosage instructions, safety information, storage conditions, and traceability. Clear labelling ensures proper use and enhances patient compliance.
Example: Blister packs with lot numbers and expiry dates prevent dosing errors and support traceability.
3. Ensure Regulatory Compliance
Packaging must meet GMP, GDP, and country-specific labeling and serialisation standards, reducing regulatory risks and preventing supply chain disruptions.
Example: Multi-dose vials with validated container-closure systems comply with FDA and EMA requirements for sterility and traceability.
4. Support Efficient Distribution
Well-designed packaging facilitates handling, storage, and transport, minimising damage and ensuring continuity across supply chains.
Example: Palletised shipments with shrink-wrap protect primary and secondary packaging during bulk transport.
5. Enhance Patient Safety and Experience
Packaging ensures that medications reach patients in optimal condition, are easy to use, and minimise administration errors.
Example: Pre-filled syringes and unit-dose sachets provide accurate dosing while reducing contamination risks.
Once the importance is clear, it helps to see how pharmaceutical packaging is structured.
3 Major Classifications of Pharmaceutical Packaging
Pharmaceutical packaging is typically classified into three levels, each serving a distinct operational and regulatory role.
| Packaging Level | Role | Key Considerations | Examples | Impact |
| Primary | Direct contact with drug; protects stability and safety | Material compatibility, CCI, E&L testing, regulatory compliance | Vials & ampoules, blister packs, bottles, sachets, pre-filled syringes | Ensures potency, sterility, and regulatory adherence |
| Secondary | Encases primary; branding, labeling, patient info | Labeling, tamper evidence, handling efficiency | Cartons, sleeves/overpacks, instruction leaflets | Protects product, supports compliance, improves usability |
| Tertiary | Bulk storage, shipping, and handling | Physical protection, temperature control, supply continuity | Palletized shipments, cold-chain packaging, multi-unit cases | Maintains integrity, prevents damage, ensures global distribution |
Now, let’s see each type of drug packaging in detail.
1. Primary Packaging: Direct Contact with the Drug
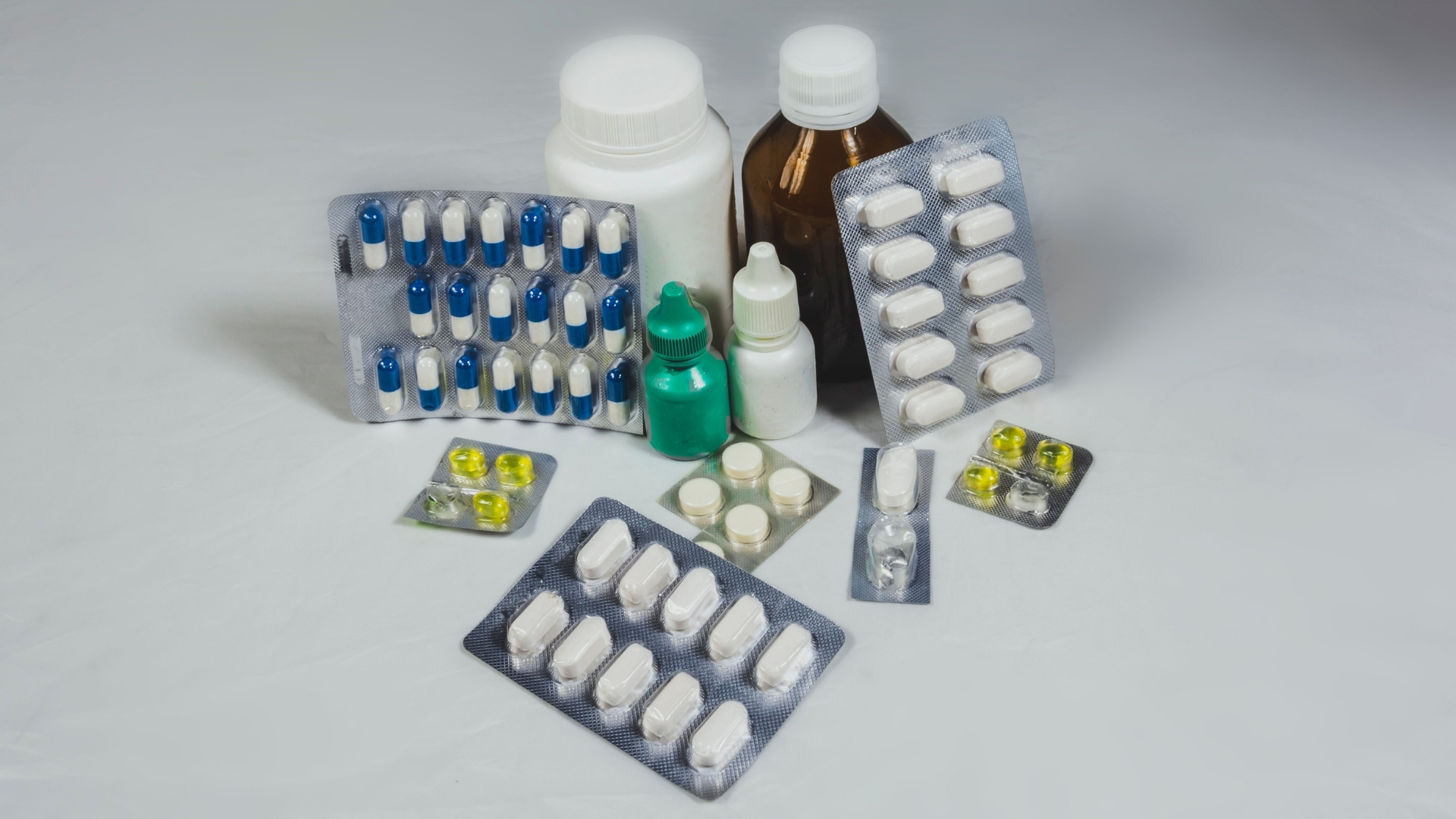
Primary packaging is the first and most critical layer of protection for pharmaceutical products, as it comes into direct contact with the drug.
Pharmaceutical companies must carefully consider material selection, extractables and leachables (E&L) profiles, and container-closure integrity (CCI), all of which are governed by strict pharmacopeial and regulatory standards.
Key Considerations:
- Material Compatibility: Glass, plastics, and metals must align with formulation properties. Example: Type I borosilicate glass maintains chemical and thermal stability for injectable biologics.
- Container-Closure Integrity (CCI): Prevents contamination, maintains sterility, and ensures product safety during storage and transport.
- Extractables & Leachables (E&L) Testing: Assesses potential chemical interactions between drug and packaging material.
- Regulatory Compliance: Ensures adherence to pharmacopeial and regional standards for accelerated approvals.
Below are the most common types and their key attributes:
a. Vials and Ampoules: Sterile Pharmaceutical Applications
Glass vials and ampoules are widely used for injectable and parenteral products, including vaccines, biologics, and high-risk formulations. Type I borosilicate glass is preferred for its chemical resistance and thermal stability, ensuring product integrity under varying storage and handling conditions.
- Container-Closure Integrity: Multi-dose vials require systems that maintain sterility throughout repeated use.
- Practical Example: A vaccine stored in a Type I vial remains chemically stable and safe for multiple doses in clinical settings.
b. Blister Packs: Unit-Dose Protection and Tamper Evidence
Blister packaging provides moisture and oxygen barriers, making it ideal for solid oral dosage forms. It also enables unit-dose dispensing, which enhances patient adherence and reduces dosing errors.`
- Clinical and Commercial Use: Commonly used for tablets, capsules, and combination therapies.
- Traceability: Supports serialization and tamper-evident features for regulatory compliance.
Example: A blister pack of antibiotics ensures each dose remains stable and traceable from manufacturing to patient use.
c. Bottles: Liquids and Solid Dosage Forms
Plastic and glass bottles are versatile containers for tablets, capsules, syrups, and suspensions. Key design considerations include:
- Child-Resistant Closures: Prevent accidental ingestion.
- Desiccant Integration: Maintains stability for moisture-sensitive products.
- Light Protection: Essential for photolabile formulations.
Example: A pediatric syrup in an HDPE bottle with a child-resistant cap ensures safe storage, accurate dosing, and extended shelf-life.
d. Sachets: Powders and Granules
Sachets offer a cost-effective, single-dose solution for powders and granules. They are particularly beneficial in public health initiatives and diverse global markets, where affordability and ease of distribution are priorities.
- Ease of Use: Single-dose format reduces dosing errors and improves patient compliance.
- Example: Oral rehydration salts packaged in sachets allow accurate dosing and simple administration in remote areas.
e. Pre-Filled Syringes and Strip Packs
Pre-filled syringes are commonly used for parenteral and biologic products where dosing accuracy, sterility assurance, and reduced handling risk are critical. From a packaging perspective, these systems require stringent container-closure integrity testing, material compatibility assessment, and validated assembly processes to ensure performance throughout storage and distribution.
- Example: A pre-filled insulin syringe ensures correct dosage, eliminates manual preparation, and enhances safety for diabetic patients.
2. Secondary Packaging: Logistics and Information Layer

Secondary packaging serves as the regulatory and informational interface of the drug product. It enables compliant labelling, tamper evidence, and traceability while protecting primary containers during handling and distribution.
From a CDMO perspective, secondary packaging must remain adaptable to region-specific labelling requirements, language variations, and serialisation mandates, particularly for products supplied across multiple regulatory jurisdictions or clinical trial regions.
Key Considerations:
- Regulatory Labelling & Patient Instructions: Packaging must clearly display batch numbers, expiry dates, dosage instructions, and storage requirements.
- Tamper Evidence & Serialisation: Supports compliance with anti-counterfeiting regulations and enhances product traceability.
- Handling Efficiency: Optimised design enables safe storage, stacking, and transportation without damaging the primary containers.
Practical Examples:
- Cartons for Oral Tablets: Secondary cartons enclosing blister packs display dosage instructions, branding, and compliance labels for retail distribution.
- Sleeves and Overpacks: Used to bundle multiple primary containers for clinical trial kits or promotional packs while providing tamper evidence.
- Instruction Leaflets: Attached leaflets in secondary packaging guide patients on administration, enhancing adherence and safety.
Impact: Effective secondary packaging protects the primary product, ensures regulatory compliance, and improves patient usability while facilitating efficient handling across logistics networks.
3. Tertiary Packaging: Transportation and Bulk Handling

For temperature-sensitive products such as biologics and vaccines, tertiary packaging must be qualified through transport simulation and stability studies to ensure temperature control throughout transit. CDMO-managed packaging programs typically integrate validated insulated shippers, temperature monitoring devices, and defined excursion management protocols.
Key Considerations:
- Physical Protection: Boxes, pallets, and shrink wraps prevent mechanical damage during storage and transportation.
- Temperature and Humidity Control: Insulated packaging and validated cold-chain solutions maintain stability for temperature-sensitive formulations such as biologics and vaccines.
- Supply Chain Continuity: Tertiary packaging supports secure transport across multiple handling points and long-distance routes.
Practical Examples:
- Palletized Shipments: Shrink-wrapped pallets for bulk tablets or vials reduce risk of impact or crushing during transit.
- Cold-Chain Packaging for Biologics: Insulated shippers with temperature monitoring protect vaccines and biologics during international shipping.
- Multi-Unit Cases: Grouping multiple secondary packages into a single tertiary package simplifies distribution and reduces handling errors.
Impact: Properly designed tertiary packaging ensures drugs reach their destination safely, maintain efficacy, and comply with global regulatory and logistical standards.
Once packaging levels are defined, material selection becomes the next critical focus.
Top 3 Materials Used in Pharmaceutical Packaging
The materials used in pharmaceutical packaging play a vital role in protecting the drug, ensuring patient safety, and maintaining regulatory compliance. Every material has unique properties that influence stability, shelf life, usability, and cost.
Choosing the right packaging material is not just about containment; it’s about preserving efficacy, supporting safe administration, and meeting global quality standards.
The table below provides a clear comparison of the most commonly used pharmaceutical packaging materials, highlighting their types, applications, and advantages:
| Material | Variants | Applications | Considerations |
| Glass | Type I (Borosilicate)
Type II (Treated Soda Lime) Type III (Soda Lime) |
Injectables, biologics, acidic or neutral aqueous products, non-parenteral products |
|
| Plastics | PET, HDPE, PP | Bottles, closures, secondary packaging |
|
| Metals / Aluminum | Aluminum foils, tubes | Blister packs, tubes for creams/ointments |
|
Now that materials are covered, let’s look at best practices that ensure performance and compliance.
6 Best Practices in the Packaging of Drugs for Stability and Regulatory Readiness
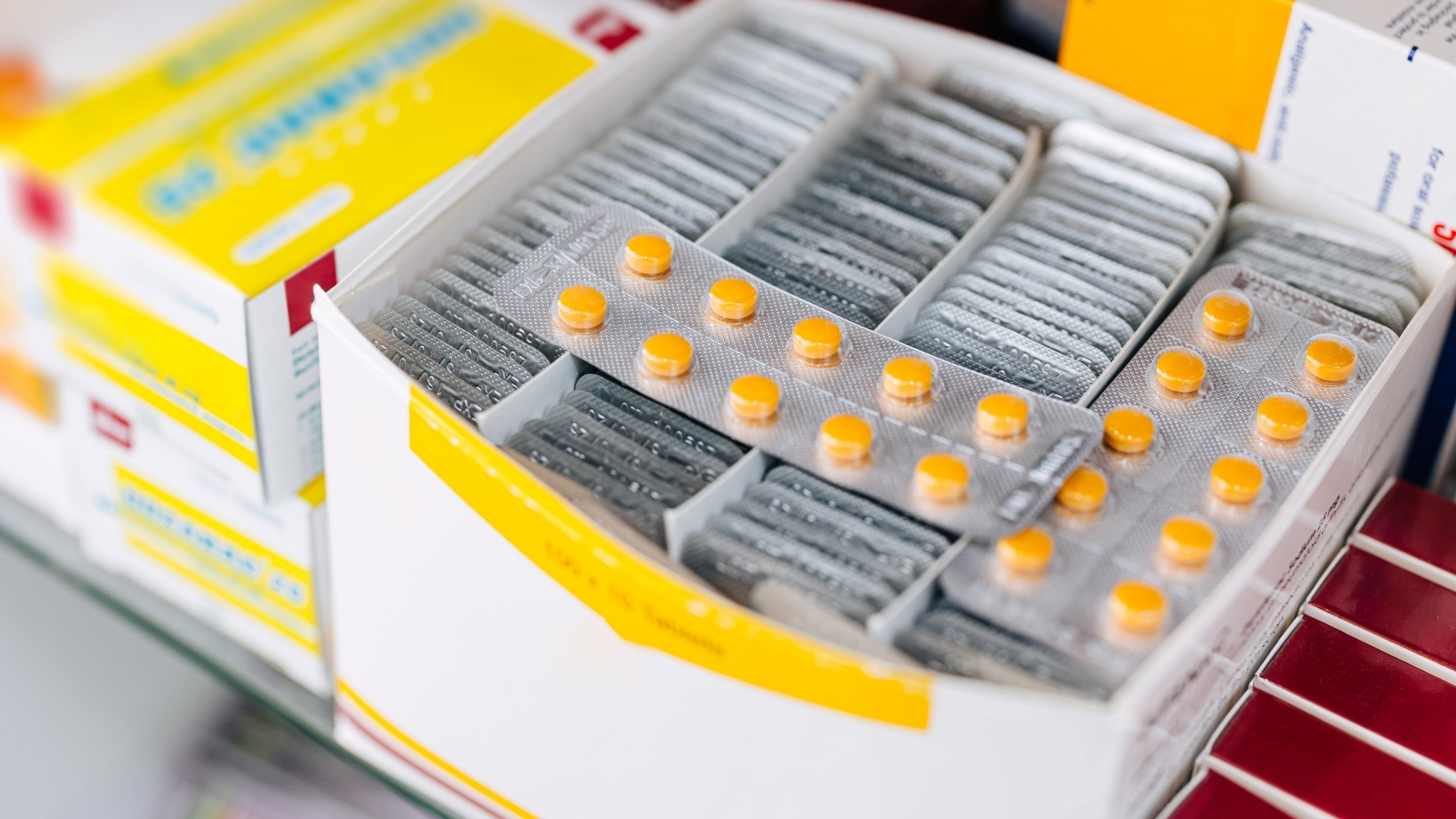
Well-designed packaging not only protects the drug but also ensures stability, usability, and traceability throughout the supply chain.
Applying these best practices helps companies reduce risk, optimise product performance, and maintain readiness for global markets.
1. Regulatory Compliance
Insight: Packaging systems must strictly adhere to GMP standards, pharmacopeial guidelines, and region-specific regulatory requirements. Comprehensive documentation, validation, and change control processes ensure consistency and inspection readiness.
Impact: Minimises regulatory risks, accelerates approval timelines, and guarantees high-quality, compliant products.
2. Product-Specific Customisation
Insight: Packaging should be tailored to the drug’s formulation, administration route, and storage conditions. Oral liquids may require airtight bottles, sterile injectables need tamper-evident vials, and moisture or light-sensitive powders benefit from high-barrier pouches.
Impact: Maintains drug integrity, improves usability, and supports patient adherence and satisfaction.
3. Sustainability Integration
Insight: Incorporating recyclable materials, reducing packaging volume, and optimising logistics helps achieve sustainability goals without compromising safety or compliance.
Impact: Reduces environmental impact, meets evolving regulatory expectations, and enhances corporate responsibility.
4. Material Compatibility and Stability
Insight: Materials must prevent chemical interactions, maintain barrier performance, and protect formulations under varying temperature and humidity conditions.
Impact: Preserves potency, extends shelf life, prevents degradation, and ensures consistent therapeutic efficacy.
5. Labelling, Traceability, and Safety
Insight: Accurate labelling, serialisation, and robust tracking systems prevent dosing errors, ensure proper usage, and support regulatory compliance.
Impact: Enhances patient safety, strengthens supply chain reliability, and enables efficient recalls when necessary.
6. Holistic, Integrated Approach
Insight: Combining regulatory compliance, product-specific customisation, sustainability, material integrity, and traceability ensures packaging is both effective and scalable.
Impact: Delivers high-quality, safe, and market-ready products while supporting operational efficiency and long-term reliability.
However, translating packaging strategies into consistent real-world performance is not without challenges.
Key Challenges in the Packaging of Drugs Across the Supply Chain
The packaging of drugs plays a critical role in maintaining product quality, safety, and compliance across the supply chain. From protecting formulations against environmental exposure to meeting evolving regulatory requirements, packaging systems must perform reliably throughout a product’s lifecycle.
The table below outlines key focus areas in pharmaceutical packaging, along with the primary considerations and their impact on product quality and regulatory compliance.
| Packaging Focus | Key Consideration | Impact on Product Quality and Compliance |
| Protection Against Contamination and Damage | Validation of container-closure systems, transport qualification studies, and ongoing stability monitoring. | Reduces risks such as breakage, seal failure, and microbial ingress, ensuring product safety and integrity. |
| Material Compatibility and Barrier Performance | Evaluation of packaging materials for chemical compatibility and moisture/oxygen barrier properties. | Maintains drug stability and potency throughout shelf life. |
| Sustainability Requirements | Incremental evaluation and adoption of sustainable materials while maintaining regulatory acceptance. | Supports environmental goals without compromising product protection or supply reliability. |
| Regulatory Compliance and Documentation | Alignment with global packaging regulations and maintenance of complete validation documentation. | Facilitates regulatory approval and minimises compliance-related delays. |
| Serialisation, Labelling, and Traceability | Use of scalable packaging systems that support region-specific labelling and serialisation requirements. | Ensures traceability, regulatory alignment, and uninterrupted supply across markets. |
| Temperature and Humidity Control | Implementation of packaging systems and storage solutions that maintain optimal environmental conditions. | Protects sensitive formulations from degradation, preserving efficacy and shelf life. |
| Tamper-Evident and Safety Features | Integration of tamper-evident seals, child-resistant caps, and protective closures. | Enhances patient safety, prevents misuse, and meets regulatory expectations. |
| Transport and Logistics Resilience | Designing packaging to withstand handling, shipping vibrations, and long-distance transport. | Minimises damage and maintains product quality from manufacturer to end-user. |
Addressing these challenges in a structured and proactive manner helps reduce risk and supports consistent market readiness.
Why CDMOs Are Central to Effective Drug Packaging?
Packaging execution becomes significantly more complex when products transition from development into multi-market clinical supply or commercial distribution. At this stage, CDMOs play a central role by integrating packaging considerations directly into formulation development, stability planning, and technology transfer workflows.
Key areas where CDMOs add value in drug packaging:
1. Packaging System Design
CDMOs help define the most suitable primary, secondary, and tertiary packaging configurations based on dosage form, stability requirements, and route of administration. Material selection considers compatibility, barrier properties, and intended market conditions to support long-term product integrity.
2. Identifying Critical Packaging Parameters
Through risk assessments and testing, CDMOs identify critical factors such as container-closure integrity, moisture and oxygen sensitivity, and light protection. These parameters ensure packaging performs reliably across storage, transport, and patient use.
3. Scaling Packaging for Commercial Supply
CDMOs manage the transition from clinical-scale packaging to commercial operations. They account for equipment capability, line speeds, serialization needs, and batch variability to maintain consistency at higher volumes.
4. Maintaining Quality and Reducing Risk
Validated packaging processes, in-process controls, and qualification studies help ensure products remain protected throughout their shelf life. Stress testing and transport simulation reduce the risk of damage, contamination, or stability failures.
5. Supporting Regulatory Compliance
CDMOs generate complete, submission-ready documentation covering packaging validation, stability data, labeling, and serialization. This supports regulatory reviews, inspections, and audits while minimizing approval delays.
A strong CDMO partner does more than package a drug. They convert packaging strategy into a robust, compliant, and scalable system that protects product integrity across development, commercialisation, and global distribution.
How DRK Research Solutions Supports Pharmaceutical Packaging?
DRK Research Solutions integrates pharmaceutical packaging considerations within its broader CDMO framework, supporting product development, clinical supply, and commercial manufacturing programs.
Packaging documentation, including compatibility assessments, stability support data, and labelling compliance, is generated as part of submission-ready development packages, reducing regulatory risk and downstream rework.
- Packaging Support: From primary container selection to secondary and tertiary packaging design, DRK provides solutions that maintain product integrity and support efficient distribution.
- GMP-Compliant Packaging Operations: Packaging activities are aligned with formulation characteristics, stability requirements, and target market regulations, ensuring consistency across development stages. Through collaborations with EU-approved manufacturing facilities in Europe and Asia, DRK supports scalable packaging execution under globally accepted quality systems.
- Expertise Across Complex Formulations: DRK supports packaging for a wide range of drug products, including oral liquids and sterile injectables, each requiring precise material compatibility and barrier performance.
- Integrated Regulatory Documentation: Packaging projects include complete validation data, stability reports, and labelling compliance documentation, facilitating submissions to global regulatory authorities.
- Flexible Packaging Solutions: Configurable packaging formats and scalable production models accommodate both clinical and commercial supply without disrupting existing processes.
APIs are sourced through qualified supplier networks, while DRK focuses on downstream drug product development, manufacturing, and packaging integration.
Through this integrated approach, DRK Research Solutions enables pharmaceutical sponsors to protect product quality, maintain regulatory compliance, and ensure market readiness through a single, trusted CDMO partnership.
Conclusion
The packaging of drugs is a critical control point across development, manufacturing, and distribution—directly influencing stability, regulatory acceptance, and supply reliability. When a packaging strategy is integrated early and executed consistently, it reduces technical risk and supports efficient market access across both regulated and emerging regions.
For sponsors managing complex formulations, multi-country clinical programs, or multi-regional supply environment supply strategies, working with an integrated CDMO partner such as DRK Research Solutions enables packaging decisions to remain aligned with development science, GMP manufacturing, and regulatory expectations throughout the product lifecycle.
For insights on optimising pharmaceutical packaging strategies and supporting efficient market readiness, connect with DRK Research Solutions for technical guidance and packaging expertise.
FAQs
1. What does pharmaceutical packaging involve?
Pharmaceutical packaging involves the materials, systems, and processes used to contain and protect drugs. It ensures product safety, maintains stability, and supports proper handling from manufacturing through patient use.
2. How is pharmaceutical packaging classified?
Pharmaceutical packaging is classified into primary, secondary, and tertiary levels. Primary packaging directly contacts the drug, secondary packaging adds protection and labelling, and tertiary packaging enables safe storage and transportation.
3. How does packaging affect drug safety and stability?
Packaging plays a critical role in protecting drugs from environmental factors such as moisture, light, oxygen, and contamination. Proper packaging helps preserve potency, extend shelf life, and maintain consistent product quality.
4. What challenges do manufacturers face in pharmaceutical packaging?
Manufacturers often deal with material compatibility issues, regulatory compliance, contamination risks, and stability concerns. Addressing these challenges is essential to ensure safe and reliable drug delivery.
5. What are considered best practices in pharmaceutical packaging?
Best practices include choosing compatible packaging materials, following regulatory standards, conducting stability and integrity testing, and using tamper-evident and protective designs to ensure drug safety and quality.
